 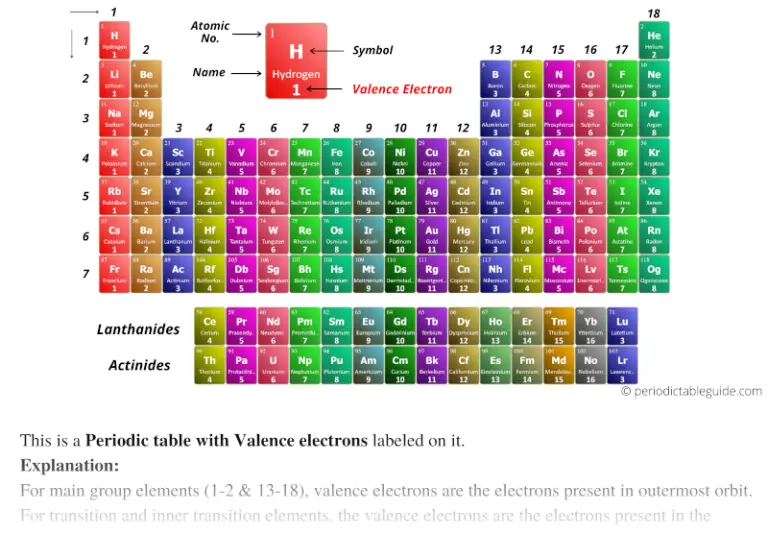
The periodic table can be divided into two broad groups:.Basically, atoms are most stable when they can achieve an outer electron structure similar to the closest Noble gas.This can be done by either sharing electrons between atoms, taking electrons from other atoms, or giving electrons away to other atoms. If atoms don't have 8 valence electrons (or two if they are close to He), then they will react with other atoms in order to have 8 valence electrons.One exception is He which has a full principle energy level with 2 electrons. All of these elements have 8 valence electrons in their highest principle energy level. One simple piece of evidence for this is the Noble Gases which form the last column on the right of the periodic table.Experimental evidence shows us that atoms are most stable when they have full s and p orbitals (8 valence electrons) in their highest principle energy level.Pick two elements from any column and determine how many valence electrons those atoms have.Bromine has 7 valence electrons: 1s 22s 22p 63s 23p 6 4s 23d 10 4p 5.Sodium has 1 valence electron: 1s 22s 22p 6 3s 1.Oxygen has 6 valence electrons: 1s 2 2s 22p 4.It is these electrons that primarily interact with other atoms. 
Valence electrons are those electrons that are in the highest principle energy level.Periodic Table and Bonding: Valence Electrons Valence Electrons
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
